![Which of the following pair of IUPAC name is not matched with its formula? (a) [Fe(H2O)s (NO)]S04: Pentaaquanitrosoniumiron(I)sulphate (b) Li[AIH4]: Lithiumtetrahydridoaluminate(III) (c) NaſAg(CN)2] : Sodiumdicyanidoargentate(1) (d) Naz[Fe(CN)s(NO ... Which of the following pair of IUPAC name is not matched with its formula? (a) [Fe(H2O)s (NO)]S04: Pentaaquanitrosoniumiron(I)sulphate (b) Li[AIH4]: Lithiumtetrahydridoaluminate(III) (c) NaſAg(CN)2] : Sodiumdicyanidoargentate(1) (d) Naz[Fe(CN)s(NO ...](https://toppr-doubts-media.s3.amazonaws.com/images/1121406/908ee0c8-a4e5-4b78-b1ec-166895164bcc.jpg)
Which of the following pair of IUPAC name is not matched with its formula? (a) [Fe(H2O)s (NO)]S04: Pentaaquanitrosoniumiron(I)sulphate (b) Li[AIH4]: Lithiumtetrahydridoaluminate(III) (c) NaſAg(CN)2] : Sodiumdicyanidoargentate(1) (d) Naz[Fe(CN)s(NO ...
![A single (6,3) sheet in the structure of [Ni(en) 2 ][Ag(CN) 2 ] 2 [Ag 2... | Download Scientific Diagram A single (6,3) sheet in the structure of [Ni(en) 2 ][Ag(CN) 2 ] 2 [Ag 2... | Download Scientific Diagram](https://www.researchgate.net/publication/244573450/figure/fig1/AS:654085460070400@1532957534231/A-single-6-3-sheet-in-the-structure-of-Nien-2-AgCN-2-2-Ag-2-CN-2-1.png)
A single (6,3) sheet in the structure of [Ni(en) 2 ][Ag(CN) 2 ] 2 [Ag 2... | Download Scientific Diagram
![Calculate the oxidation state of metal ion in: , i) `[Ag(CN)_(2)]^(-)` , ii) `[Ni(CO)_(4)]` , iii) `[Fe(CN)_(6)]^(3-)` Calculate the oxidation state of metal ion in: , i) `[Ag(CN)_(2)]^(-)` , ii) `[Ni(CO)_(4)]` , iii) `[Fe(CN)_(6)]^(3-)`](https://static.doubtnut.com/ss/web-overlay-thumb/1312614.webp)
Calculate the oxidation state of metal ion in: , i) `[Ag(CN)_(2)]^(-)` , ii) `[Ni(CO)_(4)]` , iii) `[Fe(CN)_(6)]^(3-)`
![Write IUPAC name of the following co-ordinate compounds:(i) Kleft[ Ag{ left( CN right) }_{ 2 } right] ,(ii) { K }_{ 4 }left[ Fe{ left( CN right) }_{ 6 } right] ,(iii) Write IUPAC name of the following co-ordinate compounds:(i) Kleft[ Ag{ left( CN right) }_{ 2 } right] ,(ii) { K }_{ 4 }left[ Fe{ left( CN right) }_{ 6 } right] ,(iii)](https://d1hhj0t1vdqi7c.cloudfront.net/v1/X2VUdGdsbjhBUmM=/sd/)
Write IUPAC name of the following co-ordinate compounds:(i) Kleft[ Ag{ left( CN right) }_{ 2 } right] ,(ii) { K }_{ 4 }left[ Fe{ left( CN right) }_{ 6 } right] ,(iii)
![SOLVED: Give chemical names for the following: a. [Ag(NH3)2][BF4] b. [Fe(CN) 2(CH3NC)4] c. [PtCl4]2 d. [Pt(bipy)2BrCl]2+ SOLVED: Give chemical names for the following: a. [Ag(NH3)2][BF4] b. [Fe(CN) 2(CH3NC)4] c. [PtCl4]2 d. [Pt(bipy)2BrCl]2+](https://cdn.numerade.com/ask_images/f791ec90c9d6498c8cf7414719928532.jpg)

![IUPAC name for K[Ag(CN)2] is | Coordination Master Series | Nomenclature | Cationic complex IUPAC name for K[Ag(CN)2] is | Coordination Master Series | Nomenclature | Cationic complex](https://i.ytimg.com/vi/kbSjhJsCP28/maxresdefault.jpg?sqp=-oaymwEmCIAKENAF8quKqQMa8AEB-AH-CYAC0AWKAgwIABABGFIgXChlMA8=&rs=AOn4CLCclOSVz5JNK__w96kwJCZnseiEqA)
![The IUPAC name of the complex[Ag(H2O)2][Ag(CN)2] is : | Filo The IUPAC name of the complex[Ag(H2O)2][Ag(CN)2] is : | Filo](https://classroom-images.cdn.askfilo.com/classroom/1669802704591_yxvtwsom_3268459.jpg)
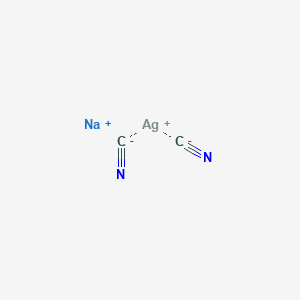
![Odia] Write the IUPAC name of the following complexes. Na [Ag (CN)2 Odia] Write the IUPAC name of the following complexes. Na [Ag (CN)2](https://static.doubtnut.com/ss/web/10790866.webp)
![The IUPAC name of the complex - [Ag(H2O)2][Ag(CN)2] is: | Filo The IUPAC name of the complex - [Ag(H2O)2][Ag(CN)2] is: | Filo](https://storage.googleapis.com/filo-classroom-notes/thumb_classroom_28484793_3E3EE.jpeg)
![Punjabi] Write IUPAC name of the following : [Ag (NH3)2] [Ag (CN)2] Punjabi] Write IUPAC name of the following : [Ag (NH3)2] [Ag (CN)2]](https://static.doubtnut.com/ss/web/12810358.webp)
![IUPAC name for K[Ag(CN)2] is IUPAC name for K[Ag(CN)2] is](https://static.doubtnut.com/ss/web/350968.webp)
![`Na[Ag(CN)_(2)]` का IUPAC नाम लिखे- `Na[Ag(CN)_(2)]` का IUPAC नाम लिखे-](https://i.ytimg.com/vi/TlQYeKdegg4/maxresdefault.jpg)
![The IUPAC name of the complex - [Ag(H2O)2][Ag(CN)2] is: | Filo The IUPAC name of the complex - [Ag(H2O)2][Ag(CN)2] is: | Filo](https://storage.googleapis.com/filo-classroom-notes/thumb_classroom_27119870_WLSJ2.jpeg)
![The IUPAC name of the complex [Ag(H2O)2][Ag(CN)2] is: A diaquasilver(I.. The IUPAC name of the complex [Ag(H2O)2][Ag(CN)2] is: A diaquasilver(I..](https://storage.googleapis.com/filo-classroom-notes/thumb_classroom_28644635_A8X1C.jpeg)


![IUPAC name for `K[Ag(CN)_2]` is IUPAC name for `K[Ag(CN)_2]` is](https://i.ytimg.com/vi/anlhlYxmO70/maxresdefault.jpg)
![Solved Na[Ag(CN)2] Sodium dicyanoargentate (I) coordination | Chegg.com Solved Na[Ag(CN)2] Sodium dicyanoargentate (I) coordination | Chegg.com](https://media.cheggcdn.com/study/c96/c9626815-93ad-47b1-8b49-69016bda3395/image.png)

![`[Ag(NH_(3))_(2)][Ag(CN)_(2)]` का IUPAC नाम लिखे- `[Ag(NH_(3))_(2)][Ag(CN)_(2)]` का IUPAC नाम लिखे-](https://i.ytimg.com/vi/EGxADHzAz9s/maxresdefault.jpg)
![The IUPAC name of the complex-[Ag(H2O)2][Ag(CN)2] is:(1) diaquasilver(II) dicyanidoargentate(II)(2) The IUPAC name of the complex-[Ag(H2O)2][Ag(CN)2] is:(1) diaquasilver(II) dicyanidoargentate(II)(2)](https://i.ytimg.com/vi/17tYTeJEp2I/hqdefault.jpg?sqp=-oaymwEmCOADEOgC8quKqQMa8AEB-AHUBoAC3AOKAgwIABABGDkgZShlMA8=&rs=AOn4CLBNP6u5qJ-XcYyY69aTT6EKhC8nfg)
![The IUPAC name of the complex - [Ag(H2O)2][Ag(CN)2] is: | Filo The IUPAC name of the complex - [Ag(H2O)2][Ag(CN)2] is: | Filo](https://classroom-images.cdn.askfilo.com/classroom/1675232308557_reobycbv_3122470.jpg)
![Write the IUPAC name of the following complexes. , `Na [Ag (CN)_2 ]` Write the IUPAC name of the following complexes. , `Na [Ag (CN)_2 ]`](https://static.doubtnut.com/ss/web-overlay-thumb/10790866.webp)
![SOLVED: Name the type of isomerism in [Ag(NH3)2][Ag(CN)2] SOLVED: Name the type of isomerism in [Ag(NH3)2][Ag(CN)2]](https://cdn.numerade.com/ask_previews/eddd20d7-9c10-4474-80bf-41e8d44699bd_large.jpg)